Secret Footage Exposes Animal Suffering in UK Drug Testing Facilities
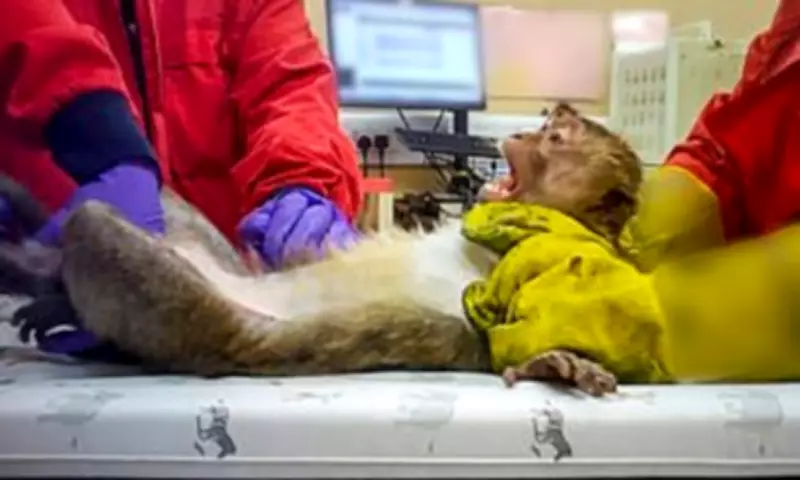
Disturbing images of monkeys having their mouths forced open and tubes pushed down their throats have emerged from secret footage filmed at two UK testing plants. The unprecedented video, supplied to The Mail on Sunday, reveals how long-tailed macaques are sacrificed to check the safety of new weight-loss medications before they can be approved for human use.
Horrifying Procedures Across Multiple Species
A former lab worker who secretly filmed the footage described being "haunted" by the shrieks and whimpers of animals during trials that could last up to two years. He revealed that beagles, pigs, rabbits, and other species also endure extreme suffering during testing for various new drugs before they reach high street chemists.
The testing covers not only potential treatments for serious diseases but also many everyday medications including headache tablets, cholesterol drugs, reflux medications, antihistamines, antibiotics, and antidepressants. All animals that survive these tests are killed at the end of the process and their bodies dissected for further studies.
Common Testing Methods Revealed
The most common procedure, called "oral gavage," involves pushing a rubber tube down the throats of restrained animals into their stomachs to feed substances directly into their bodies. This method is used for long-tailed macaques testing weight-loss drugs and liver disease medications, and for beagles testing anti-inflammatory drugs.
In other tests, masks are strapped to the faces of beagles and monkeys so they can inhale trial substances. For these procedures, monkeys are prepared by being restrained in vices around their necks and waists. Both methods were also used to test psychoactive and psychedelic compounds on beagles, including cannabis extracts and an ingredient found in ecstasy.
Additional Testing Procedures
- Mini pigs are used to test ulcer and skin infection medications by having eight cuts taken from their backs and gel applied daily
- Pregnant rabbits are used to test the effect of new drugs on embryo survival and development
- Intravenous tests involve restraining animals and injecting test compounds directly into their bloodstream
Worker's Conscience and Public Debate
The former lab worker said his conscience wouldn't let him simply quit and walk away. "I felt if I was able to provide a window into this world that had been hidden from public view, perhaps it would change," he explained. He described how procedures that the public would find shocking had been normalized as part of regulatory testing.
Campaigners have immediately called for the Government to accelerate its pledge to phase out animal testing, branding the footage "shocking." Labour MP Irene Campbell, chairman of the All-Party Parliamentary Group on Phasing Out Animal Experiments in Medical Research, stated: "The terrible suffering experienced by these animals underlines the need for bold and immediate action."
Regulatory Context and Industry Response
Both UK testing facilities are Home Office regulated and operating completely within the law. They are contracted by major pharmaceutical companies to conduct required safety tests using animals before progressing to human clinical trials.
Chris Magee from Understanding Animal Research responded that "extreme suffering" for animals in such tests was very rare and the footage likely highlighted the "rarest and most severe experiments required or permitted by law." He noted that it is already illegal to use an animal in research if a non-animal alternative is available.
Global Shift Toward Alternatives
The intervention comes after the US Food and Drug Administration announced guidance last month to help drug developers create alternatives to animal testing. The American regulator wants a shift to human-centric models that can more reliably, efficiently, and ethically predict human drug reactions prior to clinical trials.
The FDA claimed that over 90 percent of drugs that appear safe and effective in animals do not go on to receive approval in humans, predominantly due to safety or efficacy issues. They are developing replacements including computer modelling, artificial intelligence, lab-grown human organoids, and organ-on-a-chip systems.
Complexities of Complete Phase-Out
While there has been a 43 percent reduction in animals used for regulatory testing in the past decade, experts warn that stopping all animal testing won't be possible for many years. Alternatives cannot yet replicate the complexity of a full living system, which is necessary to understand how substances behave throughout an entire organism.
The debate continues between those calling for accelerated phase-out of animal testing and those who maintain that such trials remain vital for producing life-saving medication and ensuring drugs are safe for human use.