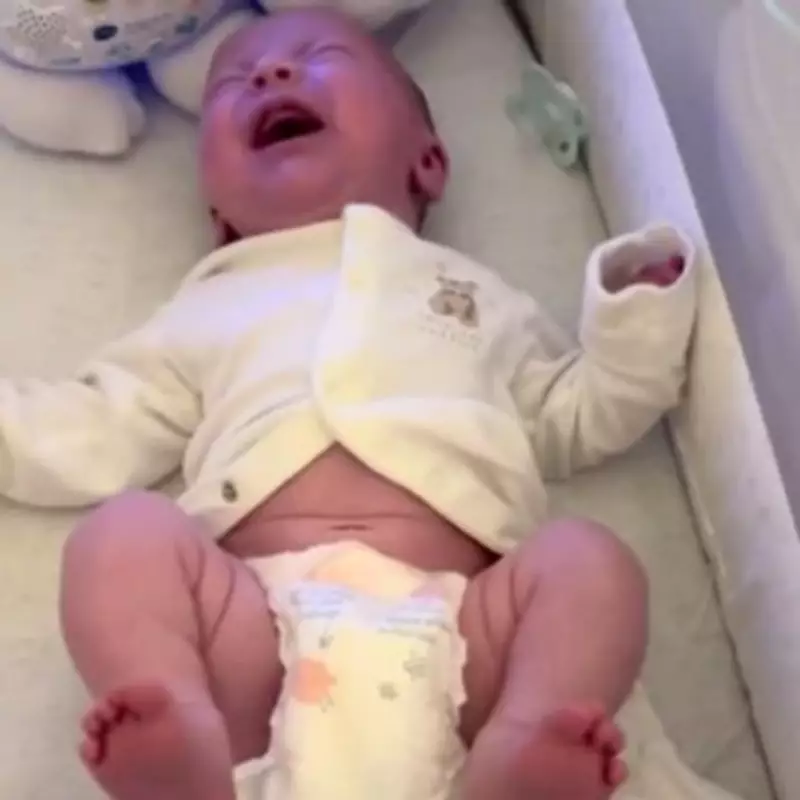
A young mother from Manchester has spoken of her profound distress after her newborn baby was hospitalised for five days, requiring oxygen and a feeding tube, following consumption of three different brands of infant formula that were later recalled due to potential toxin contamination. Catharine Alexander, a 24-year-old health and social care student, gave birth to her son Arlo Chalmers in November and initially fed him Cow & Gate First Infant Milk, which she had used previously with her first child without issue.
Rapid Deterioration and Hospital Admission
Shortly after bringing Arlo home, Catharine noticed alarming symptoms including projectile vomiting and what she described as "screaming in agony." Concerned by his rapid deterioration, she rushed the premature infant back to hospital where medical staff treated him for a suspected bacterial infection. Arlo remained hospitalised for five days while receiving oxygen support and antibiotics through a feeding tube.
Switching Formulas Without Relief
During his hospital stay, medical staff switched Arlo to SMA LITTLE STEPS First Infant Milk. However, after discharge, his symptoms persisted and even worsened, prompting Catharine to try Aptamil's First Infant Formula instead. For three consecutive months, Arlo battled daily vomiting alongside persistent diarrhoea and constipation, leaving his mother increasingly desperate for answers.
Discovery of Multiple Recalls
It was only this month that Catharine made the shocking discovery that all three formula brands she had fed her son - Cow & Gate, SMA, and Aptamil - had been subject to recall notices due to potential contamination with cereulide. This toxin, produced by certain bacteria, can cause nausea, vomiting, and abdominal cramps when consumed.
The Food Standards Agency (FSA) confirmed that specific batches from these three major formula brands might contain cereulide and advised parents who had used these products to contact their GP or NHS 111. Upon checking her supplies, Catharine confirmed the Cow & Gate batch matched recalled numbers and believes the other two brands were similarly affected.
Immediate Improvement After Brand Change
After switching to an alternative formula brand, Kendamil, Catharine reported that Arlo's symptoms disappeared "overnight." She described the transformation as dramatic, with her son becoming "a completely different baby" after just one bottle of the new formula, with no further vomiting and normal bowel movements returning immediately.
Manufacturer Responses and Safety Measures
Nestlé, manufacturer of SMA formula, confirmed they voluntarily recalled certain batches on January 5th after discovering an ingredient supplied to them did not meet quality standards. The company has discontinued sourcing from the affected supplier and is now systematically testing all ingredient batches to confirm absence of cereulide.
Danone, which produces both Cow & Gate and Aptamil, acknowledged that low levels of cereulide were identified in some specific recalled batches. Both companies have confirmed they are no longer using ingredients from the affected supplier and that products currently on sale do not contain the contaminated ingredient.
Ongoing Parental Distress and Calls for Compensation
Catharine now grapples with what she describes as persistent "mum guilt" over unknowingly feeding her infant potentially contaminated formula. She expressed anger toward the three formula brands, stating: "They've let not just me down, but all the other mothers too."
The concerned mother believes affected families should receive compensation from the formula companies and is urging other parents to carefully check batch numbers on their formula containers. She expressed particular worry about potential long-term effects on her son's digestive system after consuming what she called "toxic milk" for three months.
Medical Perspective and Official Guidance
According to health authorities, symptoms from cereulide exposure are generally self-limiting and typically resolve within 24 hours after last consumption. However, Catharine's experience suggests more prolonged effects in her infant's case.
The FSA maintains that all currently available products from these brands are safe for consumption, with contaminated batches having been removed from the market. Parents concerned about potential exposure are advised to check manufacturer websites for specific batch recall information and consult healthcare professionals if their babies display concerning symptoms.









