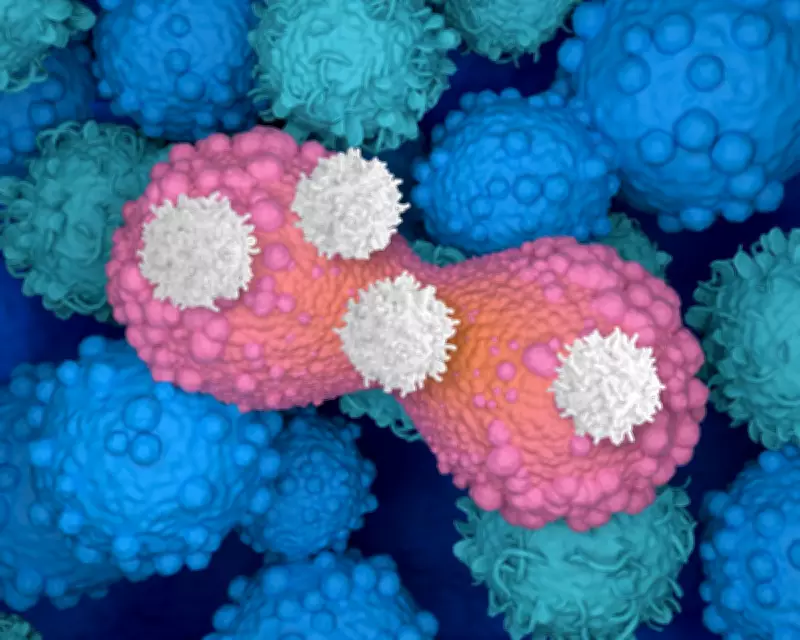
In CAR T-cell cancer treatment, T-cells are extracted from patients, genetically engineered to better hunt down cancerous cells, then infused back into the patient – and are able to prevent cancer relapses, in best-case scenarios.
The Emerging Cancer Treatment That's Exciting Scientists
After embarking on a trial of CAR T-cell therapy, actor Sam Neill announced he is cancer-free. Researchers are enthusiastic the therapies could be a major weapon in the battle against cancer.
“Game-changer.” That’s how Prof Misty Jenkins, an immunologist at the Walter and Eliza Hall Institute of Medical Research, describes CAR T-cell therapy, an emerging but still costly cancer treatment that supercharges the body’s immune system to fight disease.
Late last month, Jurassic Park actor Sam Neill put the treatment in the spotlight, revealing his stage three cancer was in remission after undergoing CAR T-cell therapy as part of a clinical trial in Sydney. He stopped short of describing his remission as a miracle – the success, he said, was “science at its best”.
The history of CAR (for “chimeric antigen receptor”) T-cell therapy is one of small discoveries accumulating over decades, leading to major advances in patient care. Pioneered in the 1990s, the therapy has exploded in the past decade. Four CAR T-cell therapies have been approved by the Therapeutic Goods Administration for use in Australia since 2018. All are for blood cancers.
The success of those therapies is increasing enthusiasm among researchers and clinicians that CAR T-cell therapies will soon become a major weapon in the battle against cancer. It is now being tweaked to combat solid tumours, with promising early signs of success tempered by the difficulties in tailoring T-cells to find their target. The future may even see it become an injectable.
“It’s such an exciting time for cancer immunotherapy, because I really feel like we’ve just scratched the surface on what’s possible,” Jenkins says.
‘Adding a GPS’ to T-cells
The immune system is dizzyingly complex. Dare to boil its functions down to a single sentence, and you could say it defends the body from foreign invasion – such as bacteria, viruses and fungi – and cleans up damaged or cancerous cells. In its arsenal are a variety of white blood cells that identify threats and remove them, including T-cells.
Cytotoxic, or “killer”, T-cells are the mercenaries of the immune system. Proteins on their surface lock onto invaders or diseased cells, such as cancerous cells, and punch holes in their target, blasting the cell apart.
But cancerous cells are adept at evading detection. CAR T-cell therapy is a way to enhance the body’s T-cell army to overcome this deficit. Jenkins describes the therapy as “adding a GPS” to the T-cells, helping the killers home in on their targets.
Typically, the therapy involves filtering a patients’ T-cells from their blood, then genetically engineering those cells outside the body to develop this GPS – the chimeric antigen receptor. The CAR T-cells are grown in the lab and then infused back into the patient, where they continue to multiply. With their enhancements they hunt and destroy their targets.
The therapy provides several advantages over traditional cancer therapies such as chemotherapy: you can make CAR T-cells specific to certain cancers and tumour types by targeting specific proteins that only cancers produce. It’s also faster, requiring only one infusion and several weeks as an inpatient compared with months-long chemotherapy regimes.
Traditional chemotherapies remain a pillar of cancer treatment, according to Dr Criselle D’Souza, an immunologist at the Peter MacCallum Cancer Centre. They’re much cheaper, treat a wider range of cancers, can be given rapidly and their side effects are well-known and so can be managed effectively.
But one huge advantage CAR T-cell therapies have is that in the best-case scenarios, the protective effect persists – preventing relapse.
“It’s like a living drug,” says D’Souza. “In theory, it can last in a patient forever if memory is created.”
She points to Emily Whitehead, the first child to be treated with CAR T-cell therapy in 2012. Her cancer has not relapsed.
‘They have to storm a formidable fortress’
Though CAR T-cell therapies have seen success in blood cancers, progress has been slower for therapies that target solid tumours like cancers of the breast, brain and prostate.
“Tackling many blood cancers is like shooting fish in a barrel compared to solid tumours, where they have to storm a formidable fortress,” says Assoc Prof Maté Biro, the laboratory head of mechanobiology at the Garvan Institute of Medical Research.
Blood cancers are susceptible to CAR T-cell therapy because the engineered cells can easily find and kill cancerous cells which circulate in the blood stream. Solid tumours, in comparison, really are a fortress. It can be hard for the immune system to access the cancerous cells in the tumour and tumours are also adept at dampening the T-cell response, evading the killers.
Researchers are developing ways to arm the T-cells with more powerful weaponry, either by developing more precise “GPS” or by supercharging their activity in different ways. There has been some early success in using CAR T-cell therapies for solid tumours, prolonging the life of those with gastrointestinal cancers and paediatric brain tumours.
“What we can also do now is start to enhance the T-cells to then also secrete drugs or secrete other antibodies into the microenvironment to help keep them alive,” says Jenkins.
There is also excitement about frontier therapies that produce CAR T-cells in vivo. Rather than removing the blood cells and engineering them in a laboratory, patients could receive an injection that helps the body generate its own CAR T-cells, in a similar way to vaccination. The in vivo treatments are just beginning to be tested in clinical trials, but Biro notes this could reduce the costs of therapy by an order of magnitude or more.
‘Hope is warranted, but so is impatience’
Cost is the most prohibitive aspect of CAR T-cell therapy today, with some treatments costing upward of half a million Australian dollars per patient. Advances like in vivo CAR T-cell therapies are expected to bring that cost down, but those are still years away.
On Tuesday, the federal government announced Carvykti, a CAR T-cell therapy for multiple myeloma, would be made freely available in public hospitals in Australia. Without funding, the cost for that treatment typically exceeds $200,000 per patient.
Access is also a problem, with Biro and Jenkins expressing concerns Australia may fall behind if it does not invest in research and sovereign capability to produce CAR T-cell therapies equitably, at home.
“These breakthroughs can only happen when the therapy expertise and infrastructure in our hospitals already exists to make and manufacture and deliver these kinds of therapies,” Jenkins says.
Neill’s high-profile “science at its best” success story provides hope. “Hope is warranted,” says Biro, “but so is impatience”.
He suggests Australia CAR T-cell therapies are a clear example of the outcomes the country could inspire if it invests in science and scientists.
“Science is delivering,” he says. “It’s the politicians and the health system that need to catch up.”









