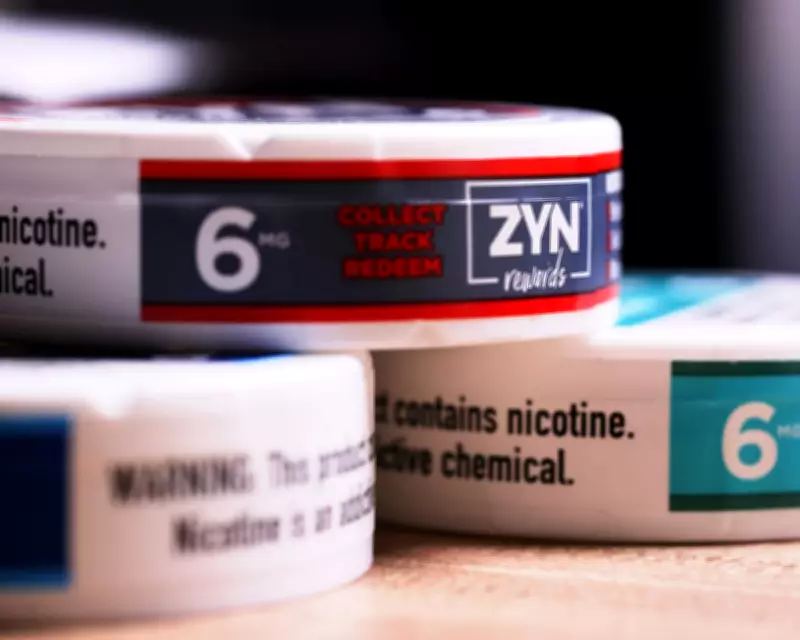
FDA Delays Authorization of Nicotine Pouches Amid Youth Addiction Concerns
The Food and Drug Administration (FDA) has not yet cleared popular nicotine pouch products for sale in the United States, despite a fast-track scheme intended to expedite approvals. Agency reviewers have raised alarms over potential risks to new users, including children, leading to significant delays in authorising brands such as Zyn and Velo.
Regulatory Hurdles and Market Impact
New tobacco products, including nicotine pouches that users insert under their lip for a nicotine buzz, must receive FDA authorisation to be legally sold in the US market, valued at approximately $22 billion. The agency evaluates whether these products can help smokers switch to less harmful alternatives while minimising risks for non-tobacco users and youth.
The previously unreported concerns behind these hold-ups have dealt a blow to major tobacco firms. Philip Morris International (PMI) has pending applications for updated versions of its top-selling Zyn label, while British American Tobacco (BAT) awaits authorisation for its rival Velo brand. Following the news, PMI's shares fell more than 7% before recovering slightly to 4.8% lower, and BAT's stock dropped 1.5%. Turning Point Brands, which also has applications in the pilot programme, saw its shares plunge over 17%.
Fast-Track Programme and Scientific Scrutiny
The FDA had planned to fast-track outstanding applications through a pilot programme, with decisions expected by the end of 2025. In December, six pouches under Altria's on! PLUS label were granted licenses. However, applications for four other pouch brands remain pending due to a cautious approach by agency reviewers. Three sources familiar with the FDA's process indicated that the scientific evidence for speeding these through was not as clear-cut as anticipated.
Specific concerns relate to risks for youth and other non-users, such as driving addiction in adults who do not already smoke. The FDA stated that nicotine products are highly addictive and not risk-free, even for adults, with nicotine potentially harming developing brains. In a statement to Reuters, the agency acknowledged that pouches are generally less harmful than cigarettes and can lower health risks when smokers switch completely, but it is carefully weighing evidence on effectiveness against risks, including survey data on pouch use among middle- and high-school students.
Industry Responses and Market Dynamics
BAT asserted in a statement that its applications warrant authorisation based on robust scientific support and that it continues to engage constructively with the FDA. PMI did not address questions about the delay but highlighted that the pilot programme could support public health and restore fair competition in the US nicotine market. Turning Point Brands declined to comment.
Jefferies analyst Andrei Andon-Ionita noted that BAT and PMI shares now heavily depend on pouches, which have become a significant part of their valuation and were previously considered less complicated by regulatory risks. The delay is particularly concerning for PMI, as Zyn has been losing market share while newer versions await licenses. Pouches are the fastest-growing nicotine product in the US, with PMI alone selling 794 million cans of its current Zyn version in 2025, more than double its 2023 sales.
Broader Context and Regulatory Pressure
The FDA's annual survey shows that nicotine pouch use among youth remains low but has risen in recent years. The pilot programme was developed amid pressure from tobacco lobbyists and the White House to authorise more brands for sale. Some manufacturers have had license applications languishing for years, leaving them unable to sell products legally or only outdated versions, while sales of unregulated nicotine products have boomed.
When asked if it was under pressure to grant more licenses, the FDA emphasised that decisions are based on science and law. The agency confirmed that applications in the pilot are on track for faster decisions than most others but declined to comment on specific concerns about youth or new user risks.









