British Scientists Pioneer Lab-Grown Oesophagus to Restore Swallowing in Breakthrough
In a landmark medical advancement, British scientists have successfully created a lab-grown oesophagus capable of restoring normal swallowing function. This breakthrough holds the potential to revolutionise treatment for infants born with severe congenital defects, particularly long-gap oesophageal atresia, where the food pipe fails to connect properly to the stomach.
Groundbreaking Research and Methodology
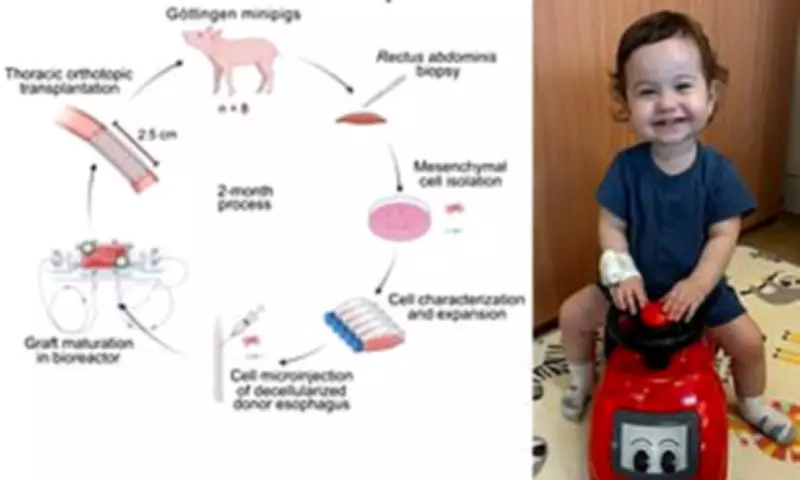
A collaborative team from Great Ormond Street Hospital and University College London engineered a replacement section of the oesophagus and utilised it in transplant operations on animal subjects. Remarkably, the procedure enabled the animals to swallow normally post-surgery. The tissue was cultivated using the recipient's own cells, eliminating the necessity for anti-rejection medications—a significant advantage as these drugs often compromise the immune system and increase infection risks.
Approximately 180 babies are diagnosed with oesophageal atresia annually in the United Kingdom. The most severe cases necessitate multiple complex surgeries shortly after birth. Without intervention, affected infants face life-threatening complications, including choking and pneumonia, due to an inability to swallow safely.
Innovative Scaffold Technique
As detailed in the journal Nature Biotechnology, researchers employed a pig's oesophagus, selected for its close resemblance to human anatomy. They meticulously stripped all living cells from the donor tissue, leaving behind a natural biological scaffold. Muscle cells harvested from the recipient animal were then introduced to this framework.
Following a week of cultivation in a specialised device, the engineered tissue was implanted. All eight animal recipients survived the surgery, resumed normal eating patterns, and maintained healthy growth rates. Over a six-month monitoring period, the lab-grown oesophagus developed functional muscles, nerves, and blood vessels, demonstrating the ability to contract and propel food towards the stomach.
Potential Impact and Future Prospects
Lead researcher Paolo De Coppi expressed optimism, stating that this work could revolutionise paediatric care within a few years. He drew parallels to the longstanding use of pig heart valves in cardiac surgery, suggesting this represents a new frontier in regenerative medicine. Dr Natalie Durkin, paediatric surgical registrar and lead author, emphasised that each step in this research marks a crucial milestone towards viable treatment options for children in the near future.
The research team believes personalised oesophagus transplants for paediatric patients could become feasible within five years. Cells could be harvested during routine procedures to grow tailor-made replacements that integrate seamlessly with the body, bypassing the need for lifelong immunosuppressive drugs.
Families' Perspectives and Expert Caution
For families grappling with this condition, the breakthrough offers a beacon of hope. Two-year-old Casey McIntyre from London, born with a segment of his oesophagus missing, has endured multiple major operations. His mother, Silviya, recounted the arduous journey, noting repeated surgeries have caused vocal cord damage. His father, Sean, highlighted the profound impact, envisioning a single early-life operation as life-changing.
However, some experts urge prudence. Professor Dusko Ilic, Professor of Stem Cell Science at King's College London, cautioned that while the study signifies a significant advance, claims of suitability for growing children are premature. He pointed out the graft's fixed length and lack of evidence for scalability with growth, alongside issues like fibrosis and stricture formation, necessitating long-term studies.
Looking Ahead
Researchers are now focused on developing longer grafts, enhancing blood supply, and preparing for initial human trials. Success could not only transform treatment for oesophageal atresia but also pave the way for repairing other organs in the future. Aoife Regan, GOSH Charity's director of impact and charitable programmes, celebrated the research success, underscoring the hope it offers to children with complex conditions and the critical role of innovative funding.









